Topcon Corporation Receives FDA Clearance for the 3D OCT-1 Maestro
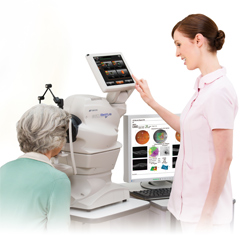
Topcon Corporation, (Headquarters: Itabashi, Tokyo Japan; President and CEO: Satoshi Hirano) (referred to as ‘Topcon’ from hereon) today announced that the U.S. Food and Drug Administration (FDA) has cleared the 3D OCT-1 Maestro and anticipates a successful launch in the U.S. market.
3D Optical Coherence Tomography is the most advanced ophthalmology diagnosis support system that acquires the cross sectional images of the posterior segment including optic nerve head and macular area. By scanning light beam across the back part of the eye, three dimensional data is collected. By analyzing the volumetric data, variety of analysis such as visualization of the retina, quantitative analysis of layer thickness, and comparison with the reference database are provided.
Additionally, the 3D OCT-1 Maestro combines with the non-mydriatic retinal photography, a simple operation with full automatic measurement and space saving installation using a flexible touch monitor. Its uniquely user-friendly design has resulted in successful adoption of the 3D OCT-1 Maestro in the most Asian, European and Middle Eastern markets. It has not only been adopted at ophthalmology clinics but has also found significant success in high-end optometric practices because of its easy operation, space-saving design and reference database comparison. Topcon anticipates that the 3D OCT-1 Maestro has massive sales potential in the U.S. market where more than 30,000 optometrists (OD) need more efficient diagnosis tools. It will be featured in American Society of Retina Specialists (San Francisco.) in August and will be available for sale by Topcon Medical Systems, Inc.
About Topcon Medical Systems, Inc.
http://www.topconmedical.com/






